The Fda Regulates Facilities That Manufacture Otc Medications.

Hiring An Fda Registered Facility For Supplement Manufacturing


An Overview Of Fda Requirements For Otc Drugs Over The Counter Products And Understanding Fda Regulation For Otc Drugs Meet All The Fda Requirements Quick And Easy Excellent Customer

What You Need To Know About Manufacturing Otc Products

What You Need To Know About Manufacturing Otc Products

What You Need To Know About Manufacturing Otc Products

Fda Requirements For Hand Sanitizers And Other Antiseptic Otc Drugs Fda Listing Inc

Securing The U S Drug Supply Chain Oversight Of Fda S Foreign Inspection Program 12 10 2019 Fda

How Fda Regulates Hand Sanitizers

1 Otc Supplement Manufacturer Speedy Quotes Ssmfg
![]()
Fda Update New U S Legislation To Impact The Over The Counter Otc Drugs Industry

Securing The U S Drug Supply Chain Oversight Of Fda S Foreign Inspection Program 12 10 2019 Fda

Covid 19 And Beyond Oversight Of The Fda S Foreign Drug Manufacturing Inspection Process 06 02 2020 Fda

Securing The U S Drug Supply Chain Oversight Of Fda S Foreign Inspection Program 12 10 2019 Fda

Securing The U S Drug Supply Chain Oversight Of Fda S Foreign Inspection Program 12 10 2019 Fda
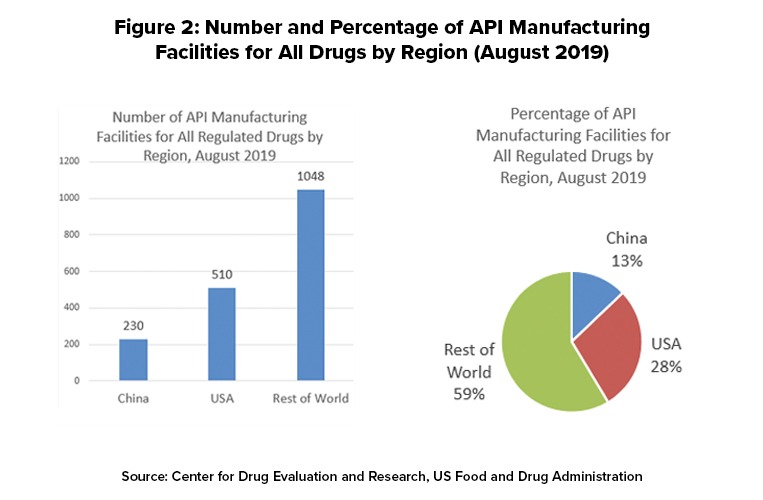
Api Sourcing The Supply Side For Us Marketed Drugs Dcat Value Chain Insights
Otc Drug Compliance Solutions With The Fda Din Number

What You Need To Know About Manufacturing Otc Products

Fda Announces Facility Fees For Otc Drug Manufacturers Sheppard Mullin Richter Hampton Llp Jdsupra
Fda Requirements For Hand Sanitizers And Other Antiseptic Otc Drugs Fda Listing Inc

Comments
Post a Comment